|
Then come days of checking and quality assurance in which the DNA material is tested to ensure it meets the strict requirements of the vaccine. The bags can’t fail, because each contains the building blocks of the COVID-19 vaccine. Pfizer must vet the bag producer and check every lot that comes in, so it can verify they meet federal manufacturing guidelines. The lines of DNA in the now-clear solution are then packed into special high-tech bags about the size of a grocery bag and frozen to minus 112 F for storage. 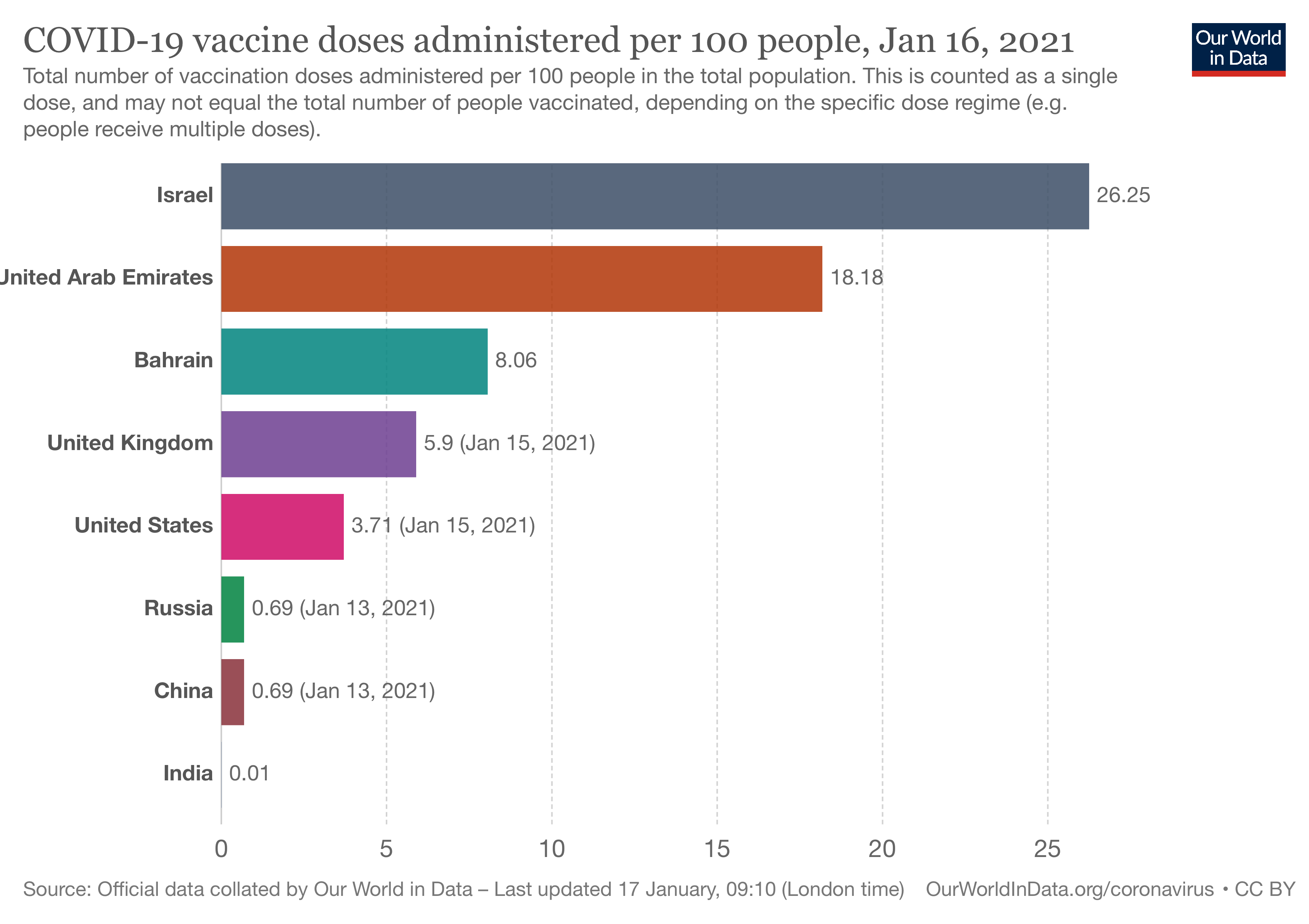
Next, the loops are straightened out, or "linearized," using enzymes to cut the circles. When completed, the solution goes through a purification process – akin to straining spaghetti through a colander – that removes everything but the microscopic loops of DNA. "When the FDA licenses a vaccine, they license the building, they license the product and they license the process," he said. Paul Offit, director of the Vaccine Education Center at Children's Hospital of Philadelphia. “We don’t have recalls," he said. "Nothing problematic could get out the door because it’s such a strict regulatory process.”īoth the end product and the production process are heavily regulated by the Food and Drug Administration, said Dr. Vaccines are given to healthy people, so companies must err on the side of caution, Van Exan said. More than half of the production time for Pfizer's COVID-19 vaccine is devoted to testing and quality assurance – making sure the resulting product, at each stage, is safe, pure and exactly the same as the tested vaccine that proved effective. 
The Salk polio vaccine takes a full 18 months to make, said Robert Van Exan, a 40-year vaccine industry veteran who now does consulting out of Ontario, Canada. Still, vaccine arriving at hospitals and clinics today comes from batches started just before Halloween.Įven at the current 110 days, Pfizer's COVID-19 vaccine production time is more than two months faster than that of the influenza vaccine. The time per batch from start to finish was originally about 110 days, but as engineers learn how to make the process more efficient, the company thinks it will get down to just 60 days per batch. But no matter how much engineers like Calitri have worked to shave weeks, days and hours off the vaccine-making process, it still takes time.įor Pfizer-BioNTech, a batch can make anywhere from 1 to 3 million doses of vaccine per production run, which the company says will soon take just 60 days. "It’s not like (making) orange juice."Īll the competitive COVID-19 vaccine manufacturers have been pushing to produce more doses faster. "People don’t understand, manufacturing vaccines is extremely complicated," AstraZeneca CEO Pascal Soriot said at a recent news conference. Now, the company is struggling to meet its production commitments in Europe, because starting up a factory in Belgium proved harder than anticipated. Not enough older people were included to make it clear whether they'd be protected. Merck, another pharmaceutical giant, dropped out of the race recently when its early vaccine candidates failed to live up to their promise.Īnd AstraZeneca, which is collaborating on a vaccine with Oxford University in England, has stumbled a few times. In a global trial, effectiveness figures were muddied by giving some participants a different vaccine dose. 11.īut the vaccine development and manufacturing process doesn't always go smoothly. The two teams, both of which use a technology called mRNA, have delivered more than 59 million doses to the federal government since Dec. Pfizer, which partnered with a German company, BioNTech, had a 44,000-person clinical trial finished before Thanksgiving. Moderna had a vaccine ready to be tested in people in just over two months. 
The two vaccines rolling out across the country since late December have made the vaccine development and production process look easy. "It is science with established principles, but sometimes is more idiosyncratic than art." There's variability in the raw materials, the microorganisms needed to grow vaccine products, the conditions of the culture in which those microorganisms are grown, and more, Yadav said. Plasmid manufacturing suite at Pfizer's Chesterfield, Mo., site.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
